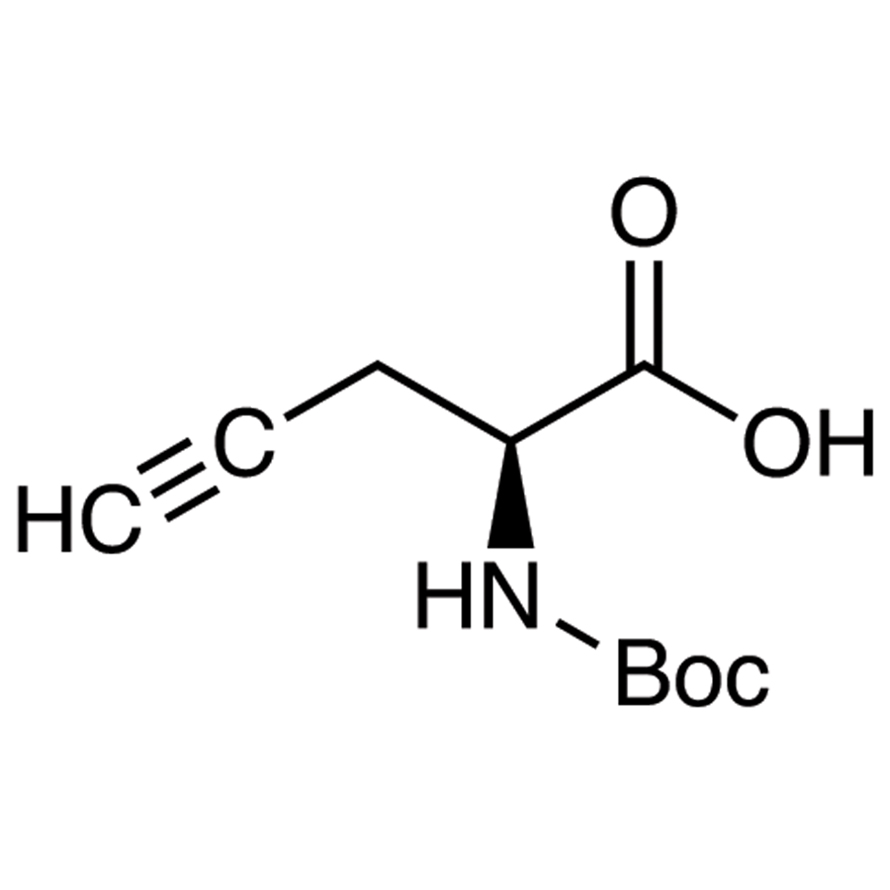
Expedited synthesis of α-amino acids by single-step enantioselective α-amination of carboxylic acids | Nature Synthesis
Boc-L-Glutamic acid, 5 g, CAS No. 2419-94-5 | tert-Butyl / Boc | Amino acids, protected | Amino Acid Derivatives | Amino Acids and Amino Acid Derivatives | Organic & Bioorganic Chemicals | Chemicals | Carl Roth - International

293302-31-5 (net) | Bis-Boc-amino-oxyacetic acid monohydrate | = 98% (HPLC) - Chem-Impex International

An efficient and highly chemoselective N-Boc protection of amines, amino acids, and peptides under heterogeneous conditions | SpringerLink

Synthesis of All Nineteen Appropriately Protected Chiral α-Hydroxy Acid Equivalents of the α-Amino Acids for Boc Solid-Phase Depsi-Peptide Synthesis | Organic Letters
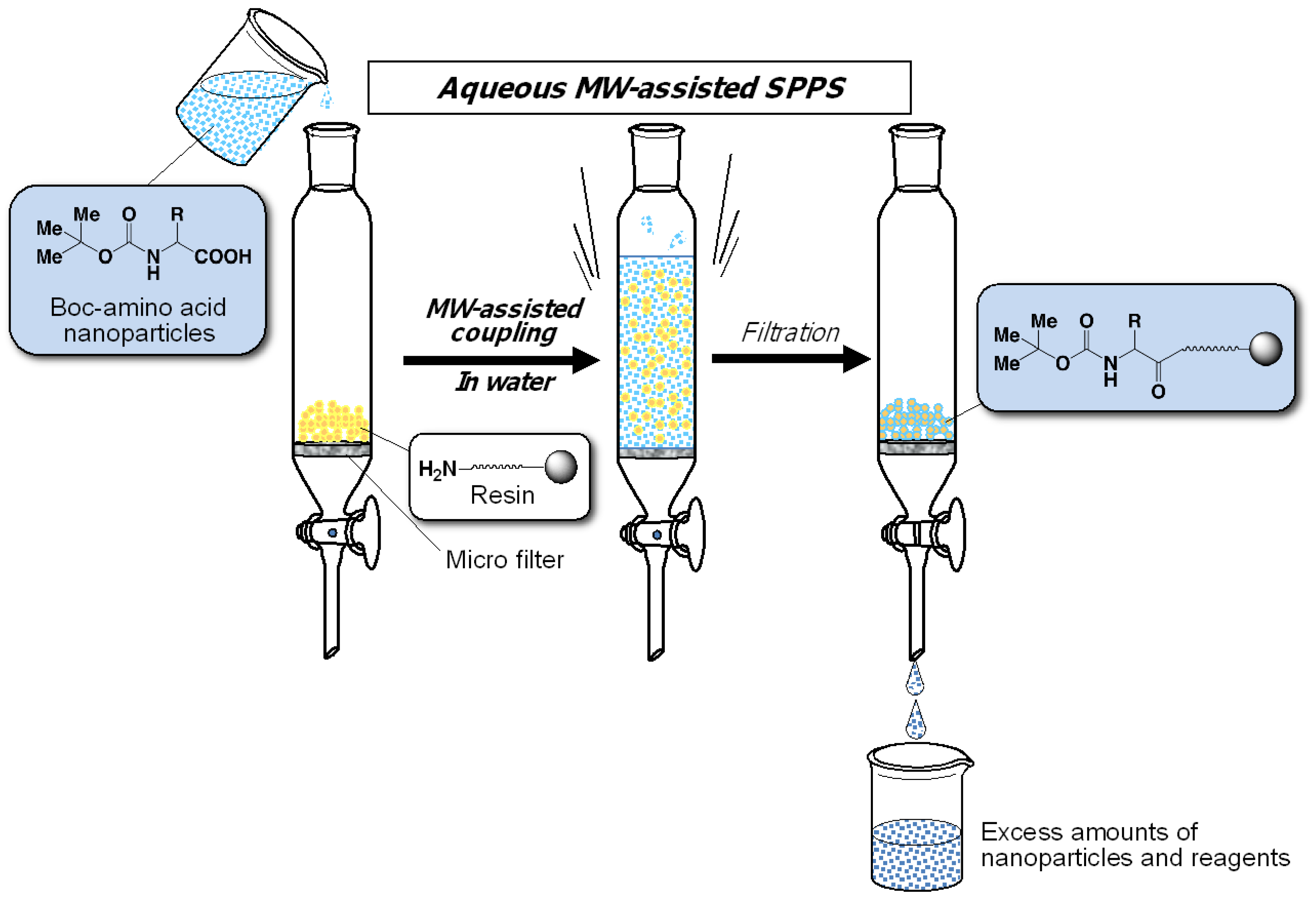
Applied Sciences | Free Full-Text | Aqueous Microwave-Assisted Solid-Phase Synthesis Using Boc-Amino Acid Nanoparticles

Direct amidations between N-Boc protected β-amino acid and C-protected... | Download Scientific Diagram

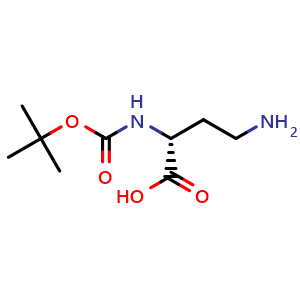





![Synthonix, Inc > 90495-99-1 | 2-[[2-(Boc-amino)ethyl]amino]acetic Acid Synthonix, Inc > 90495-99-1 | 2-[[2-(Boc-amino)ethyl]amino]acetic Acid](https://synthonix.com/images/product/74883.png)