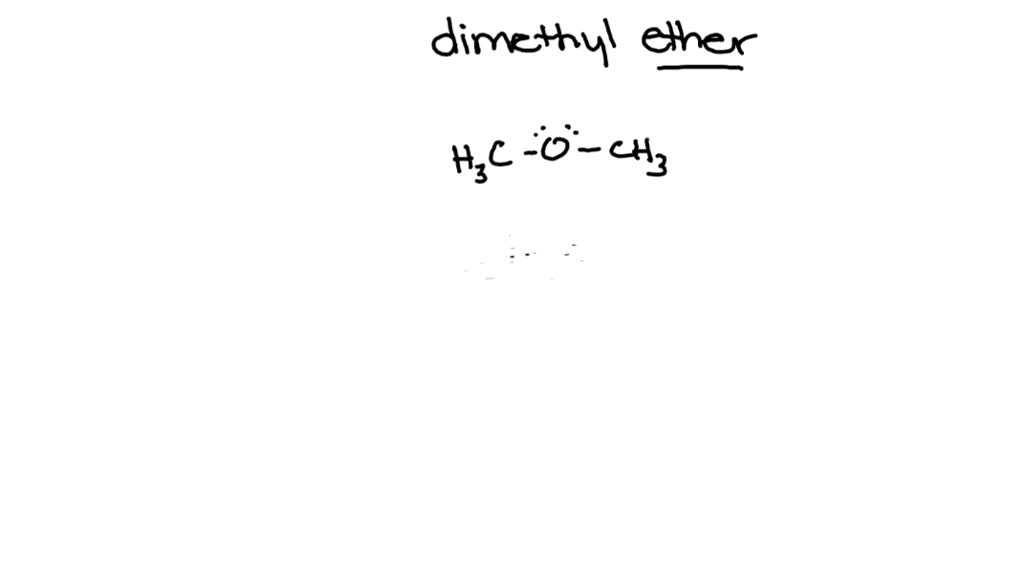
SOLVED: Draw the 3D structure of Dimethyl Ether (C2H6O). Indicate the shape and the corresponding bond angles. Indicate if the molecule is polar or nonpolar.

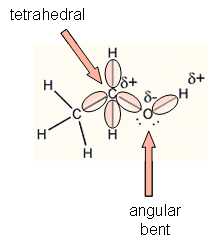
Is CH2O Polar or Nonpolar? - Polarity of Formaldehyde | Molecular geometry, Electron configuration, Polar

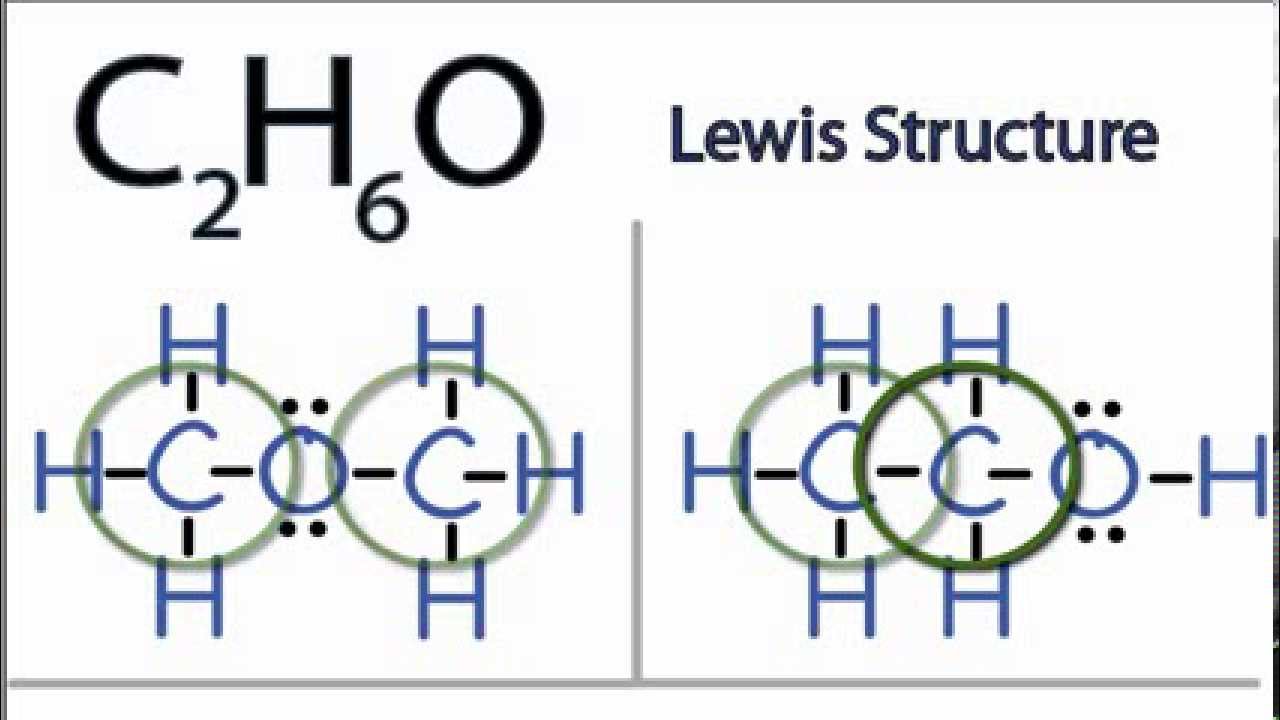
SOLVED: a. There are two forms of C2H6O, one of which is a liquid at room temperature and the other is a gas. (More than one structure for the same molecular formula

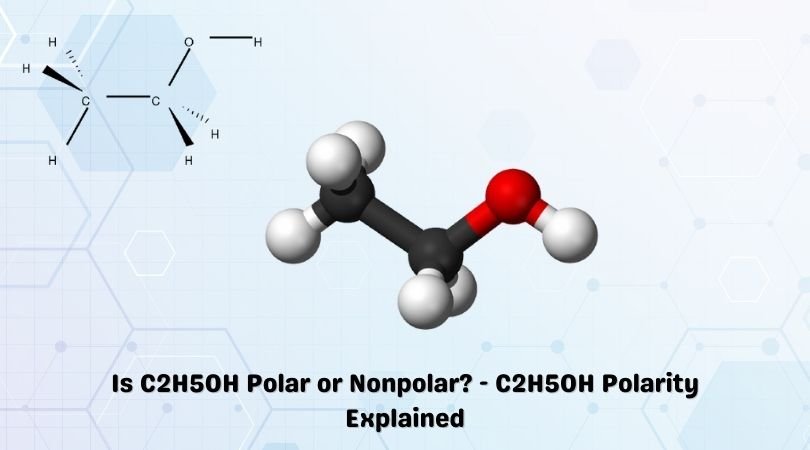
Using an arrow, indicate the overall direction of the dipole moment of C2H6O. Determine if there is more than one isomer for the given molecular formula. If so, draw them. | Homework.Study.com











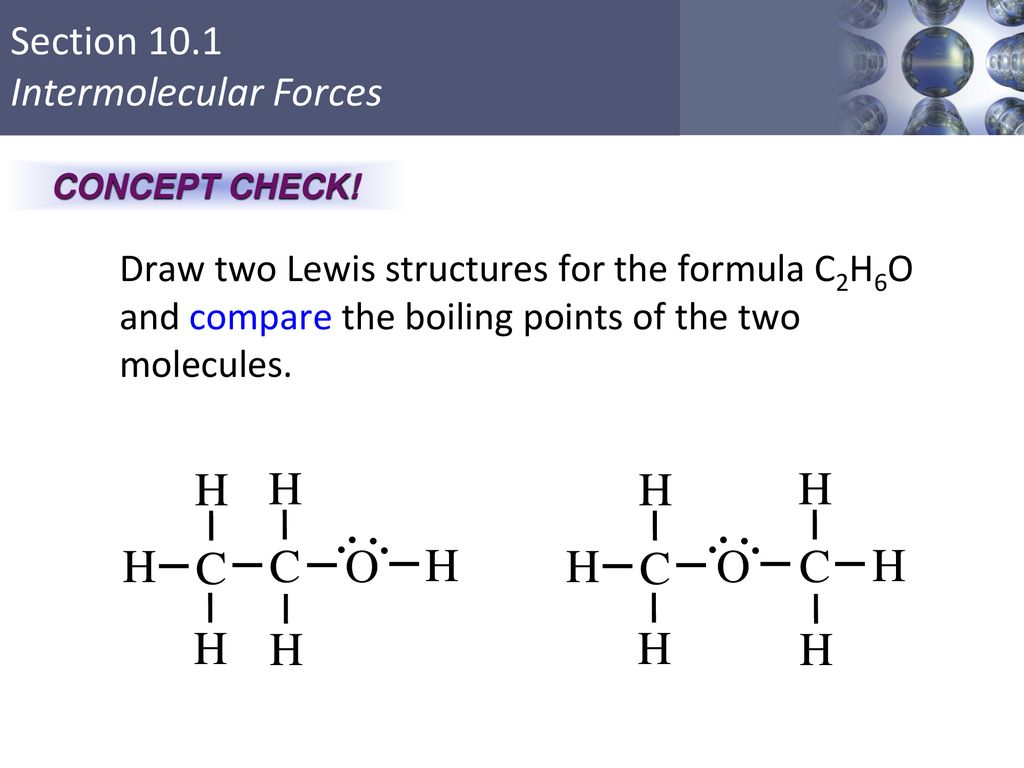
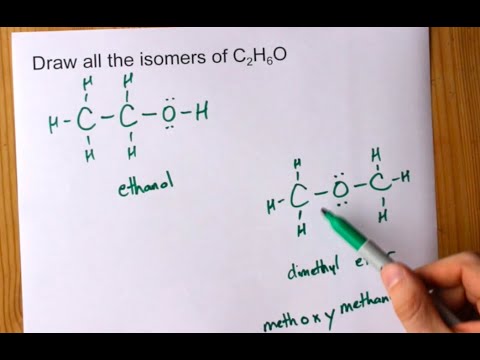


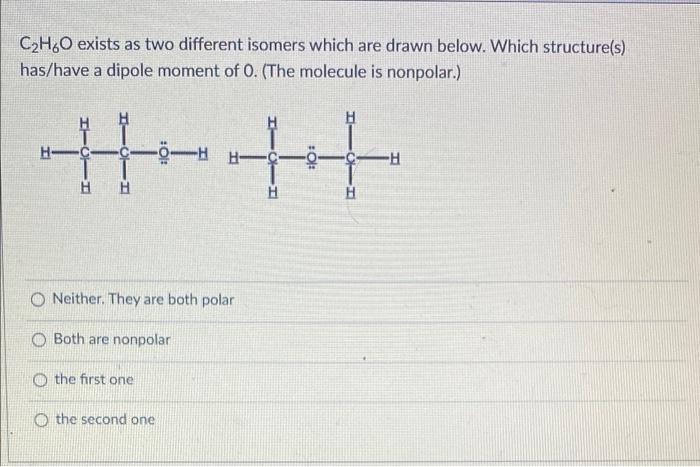

![Biology] Polar Bonds - General Discussion - Neowin Biology] Polar Bonds - General Discussion - Neowin](https://cdn.neowin.com/forum/uploads/post-107847-1188869267_thumb.jpg)
