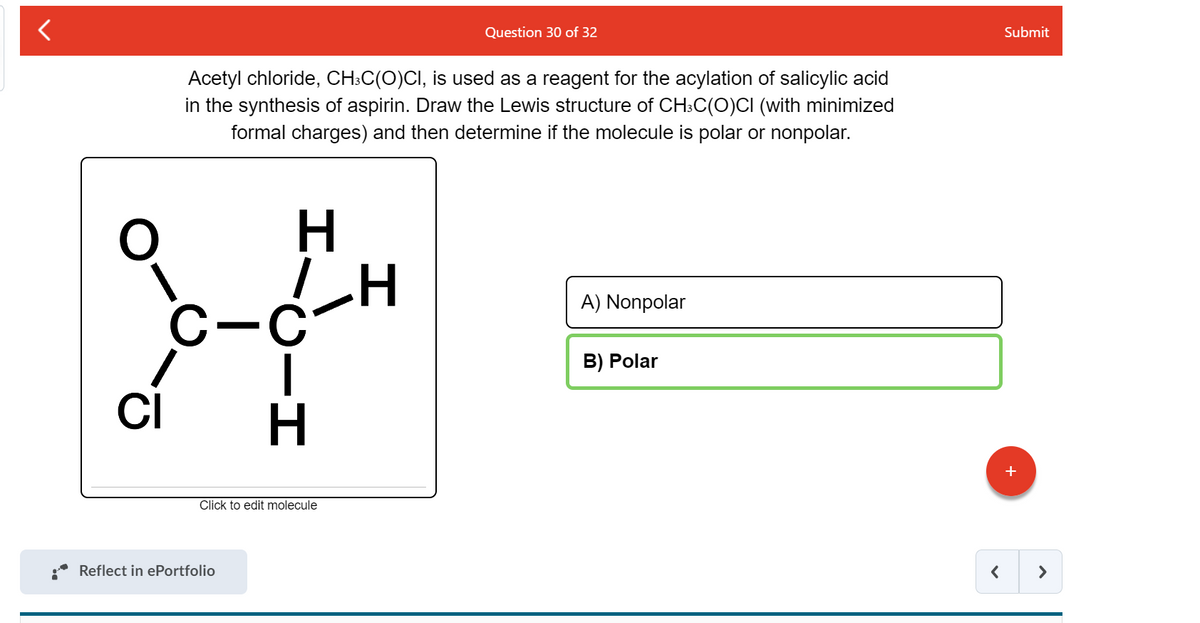
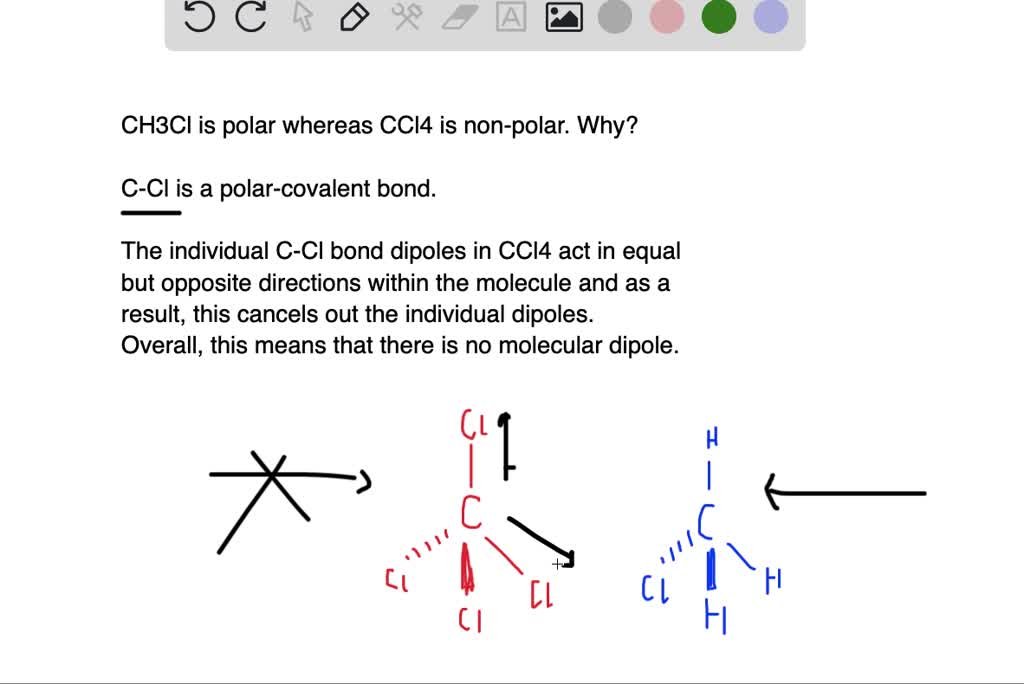
Cambridge International AS and A Level Chemistry Coursebook with CD-ROM by Cambridge University Press Education - Issuu
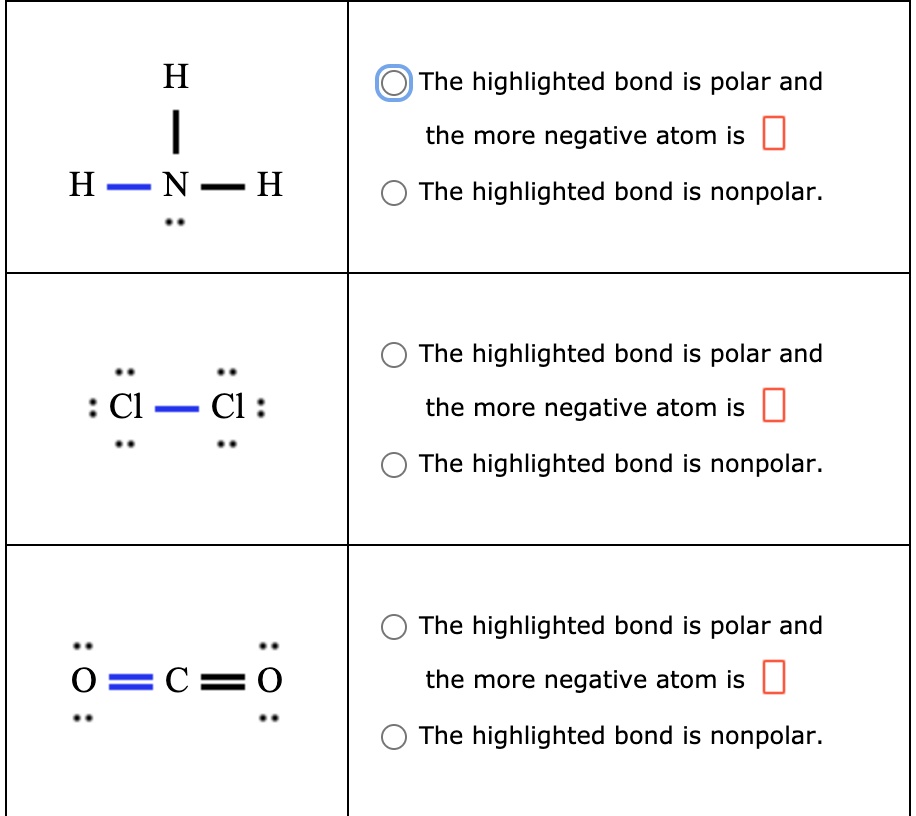
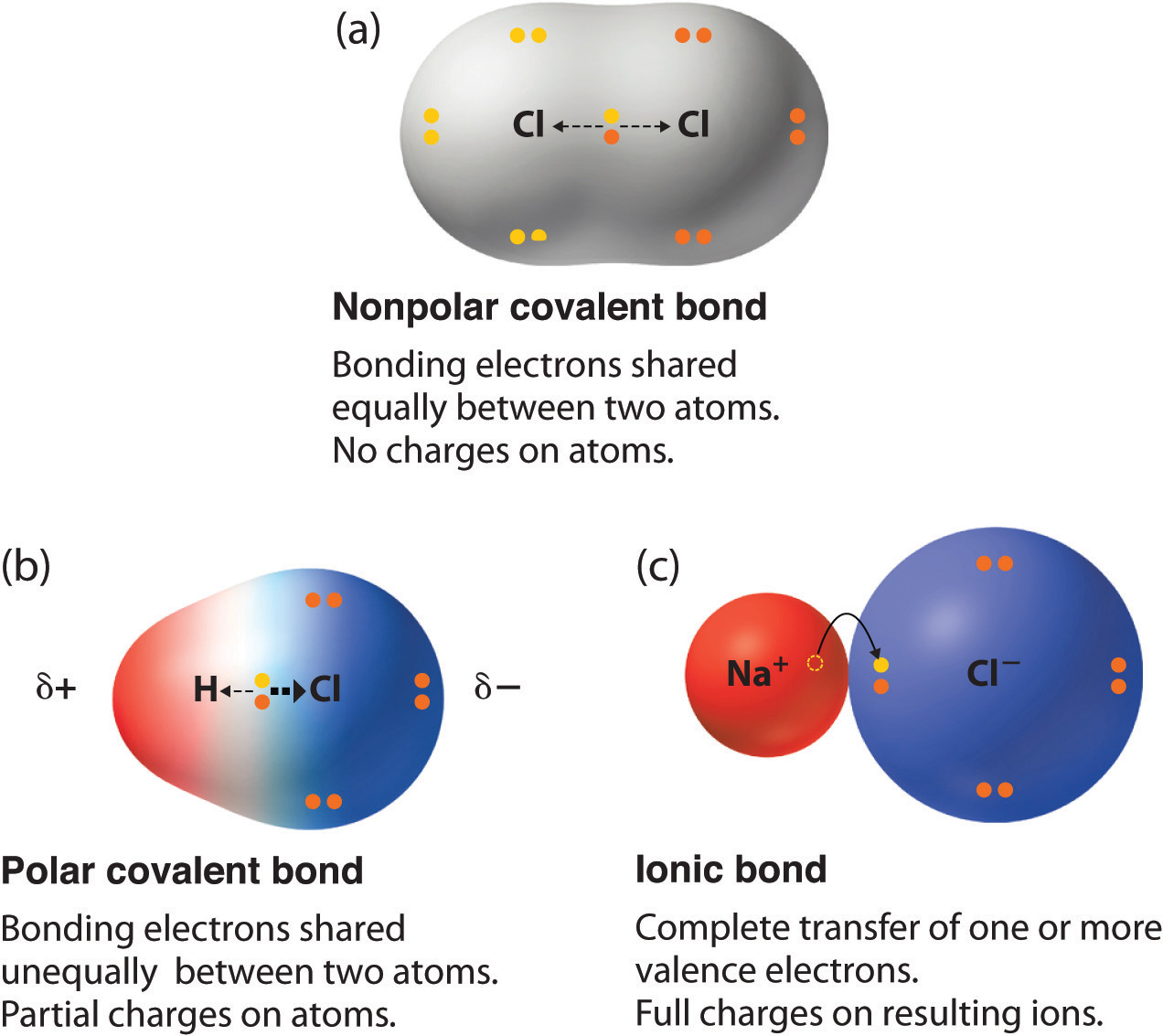
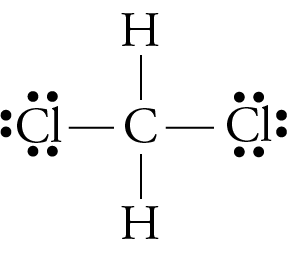
SOLVED: H The highlighted bond is polar and the more negative atom is HNH The highlighted bond is nonpolar. The highlighted bond is polar and the more negative atom is Cl Cl :
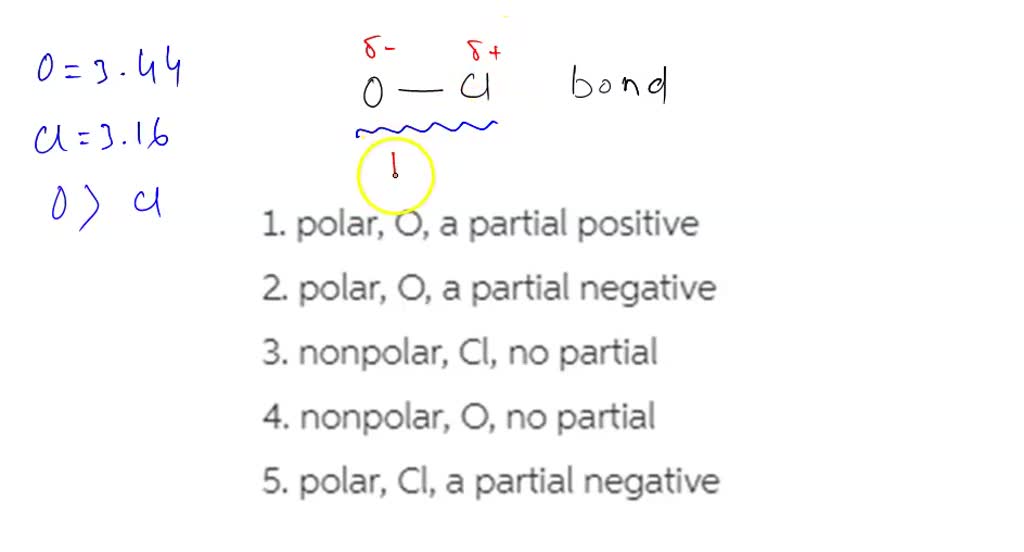
SOLVED: In an oxygen-chlorine bond, the bond is polar and the O atoms bear a partial negative charge.

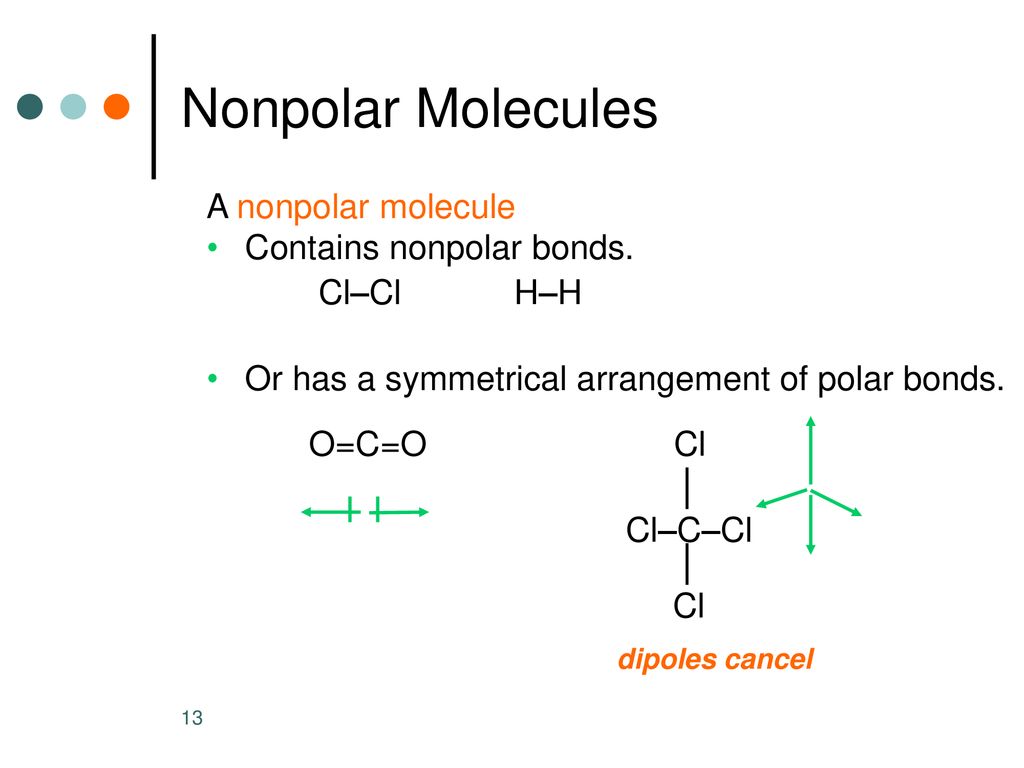
![Best Overview: Is BeCl2 Polar or Nonpolar [No#1] - Science Education and Tutorials Best Overview: Is BeCl2 Polar or Nonpolar [No#1] - Science Education and Tutorials](https://sciedutut.com/wp-content/uploads/2021/05/Is-BeCl2-Polar-or-Non-Polar-2-1024x493.png)