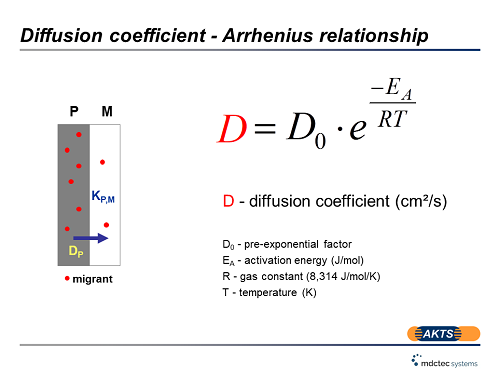
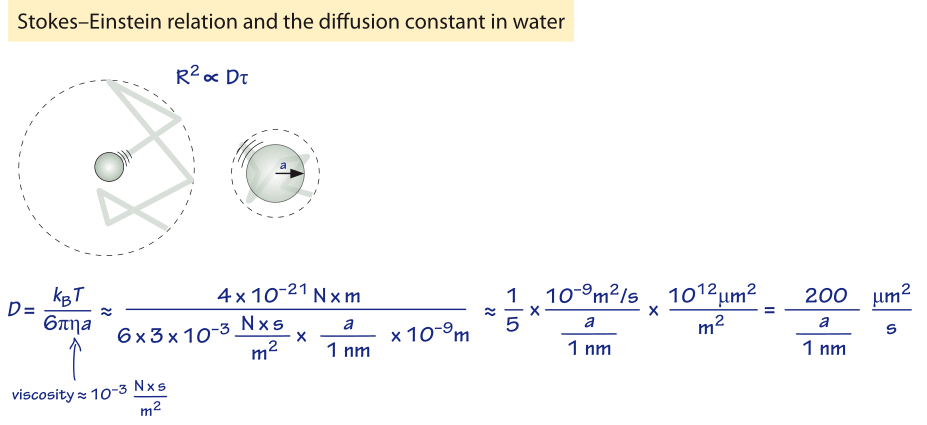
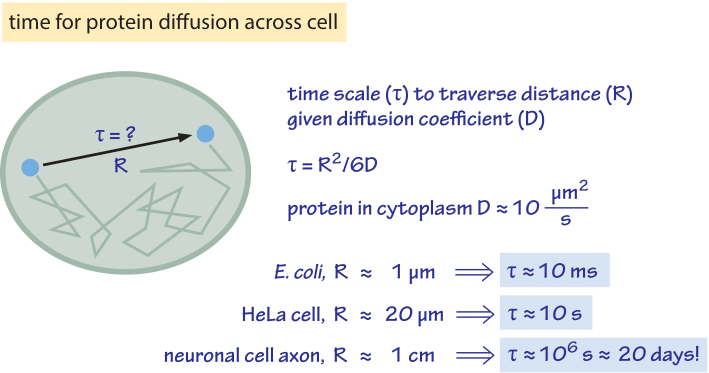
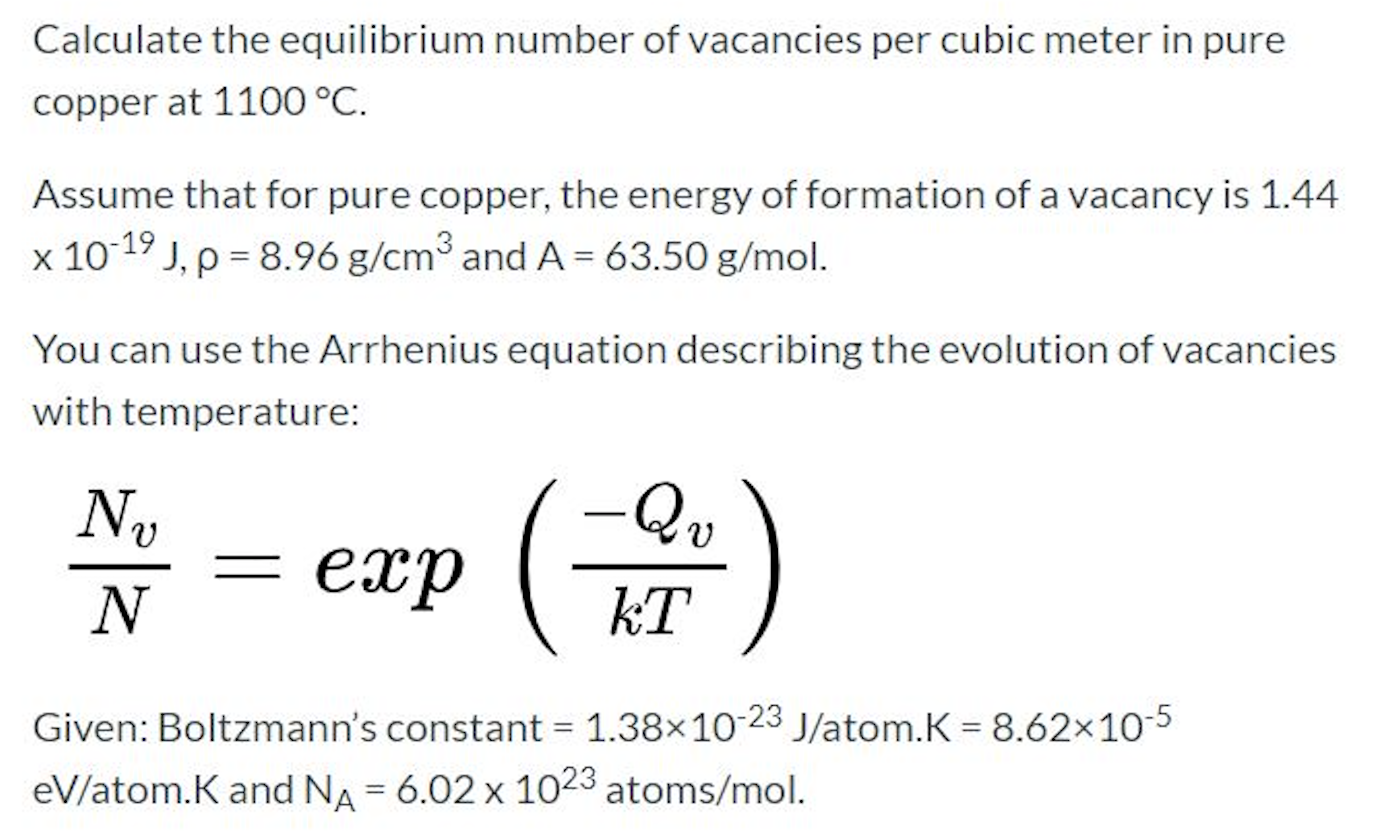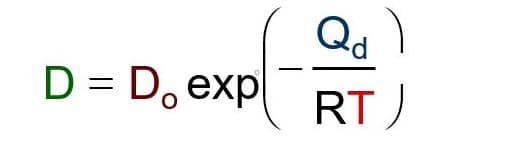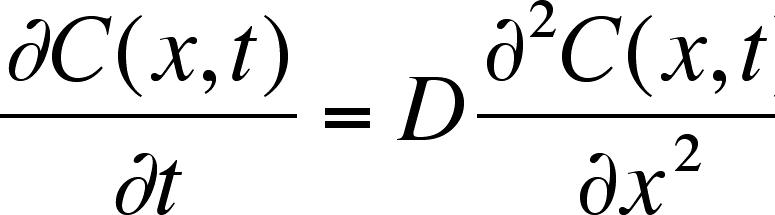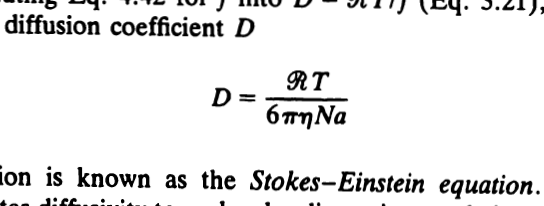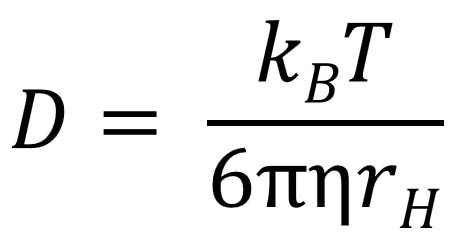Quantitative Interpretation of Protein Diffusion Coefficients in Mixed Protiated–Deuteriated Aqueous Solvents | The Journal of Physical Chemistry B
![SOLVED: Distinguish between a Diffusion Coefficient and a Mean Square Displacement. [4 marks] Why is the correct value for viscosity so important in calculating the diffusion coefficient? [2 marks] Calculate, using the SOLVED: Distinguish between a Diffusion Coefficient and a Mean Square Displacement. [4 marks] Why is the correct value for viscosity so important in calculating the diffusion coefficient? [2 marks] Calculate, using the](https://cdn.numerade.com/ask_images/2cc00b0431fa47618b854c7d2fa8986b.jpg)
SOLVED: Distinguish between a Diffusion Coefficient and a Mean Square Displacement. [4 marks] Why is the correct value for viscosity so important in calculating the diffusion coefficient? [2 marks] Calculate, using the

Simple and accurate correlations for diffusion coefficients of solutes in liquids and supercritical fluids over wide ranges of temperature and density - ScienceDirect

SOLVED: According to the Kinetic Theory of gases, the diffusion coefficient; D, of a gas can be described using the following equation: 1 D = v^ 3 Using this ratio calculates the
ACP - Viscosities, diffusion coefficients, and mixing times of intrinsic fluorescent organic molecules in brown limonene secondary organic aerosol and tests of the Stokes–Einstein equation

OneClass: The diffusivity coefficient of a substance can be calculated from Fick’s Law and is repre...