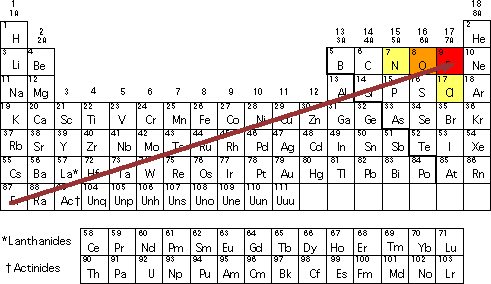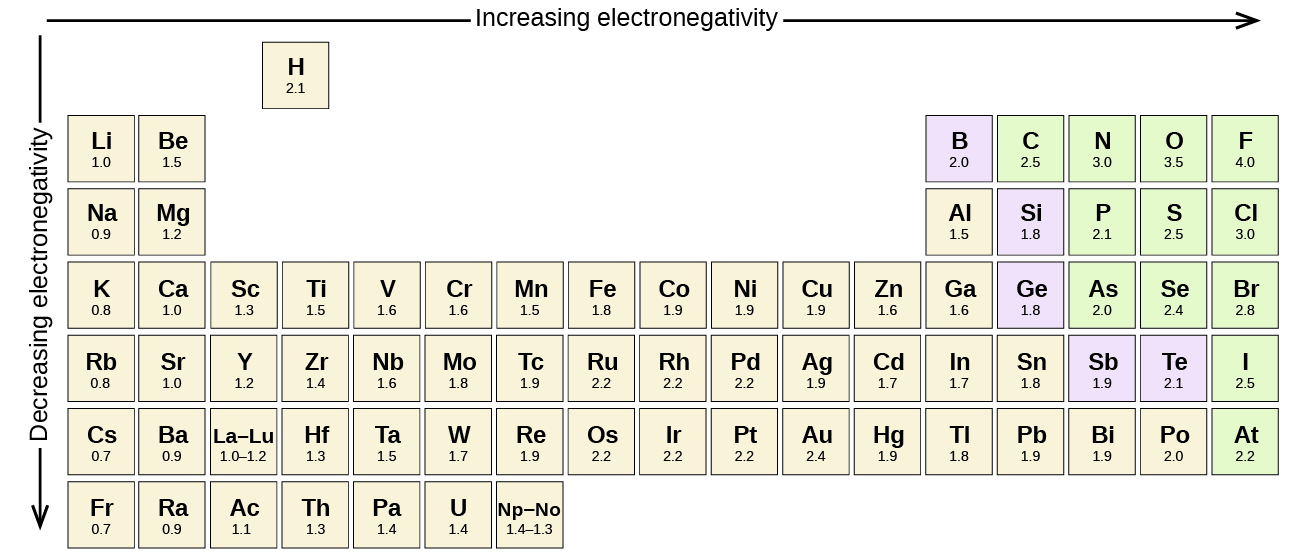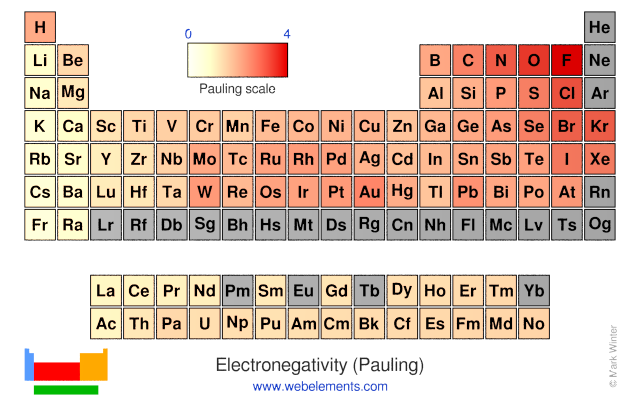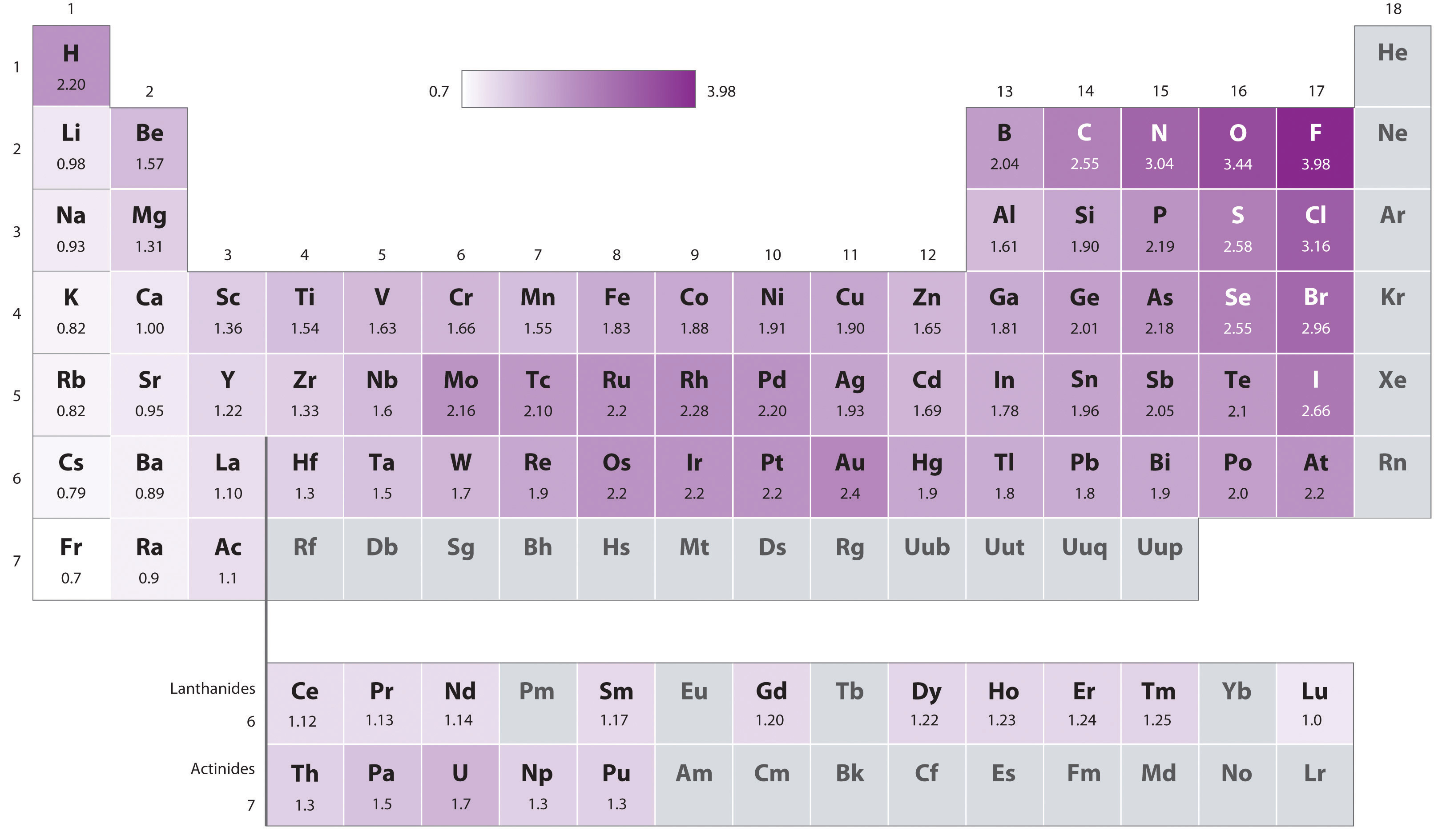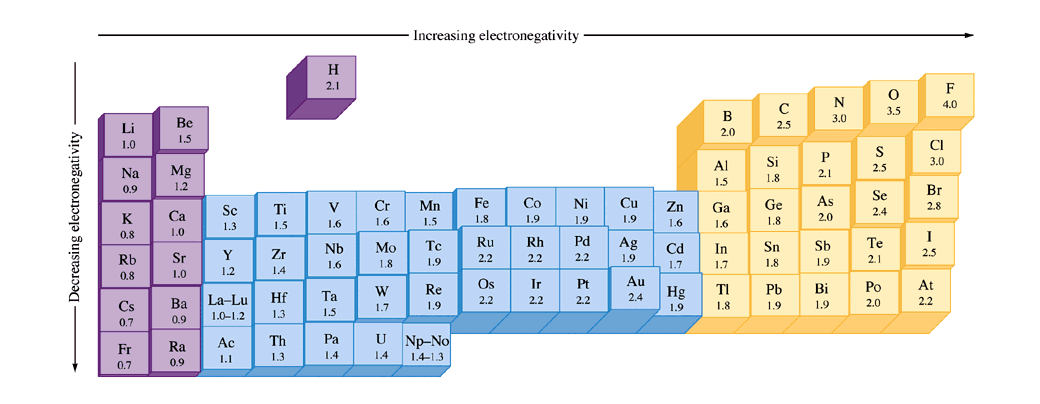
What trend in electronegativity do you see as you go down a group/family on the periodic table? | Socratic

SOLVED:Judging from their relative positions in the Periodic Table, which element in each pair has the larger electronegativity? (a) F or Cl (b) O or S (c) C or N (d) C