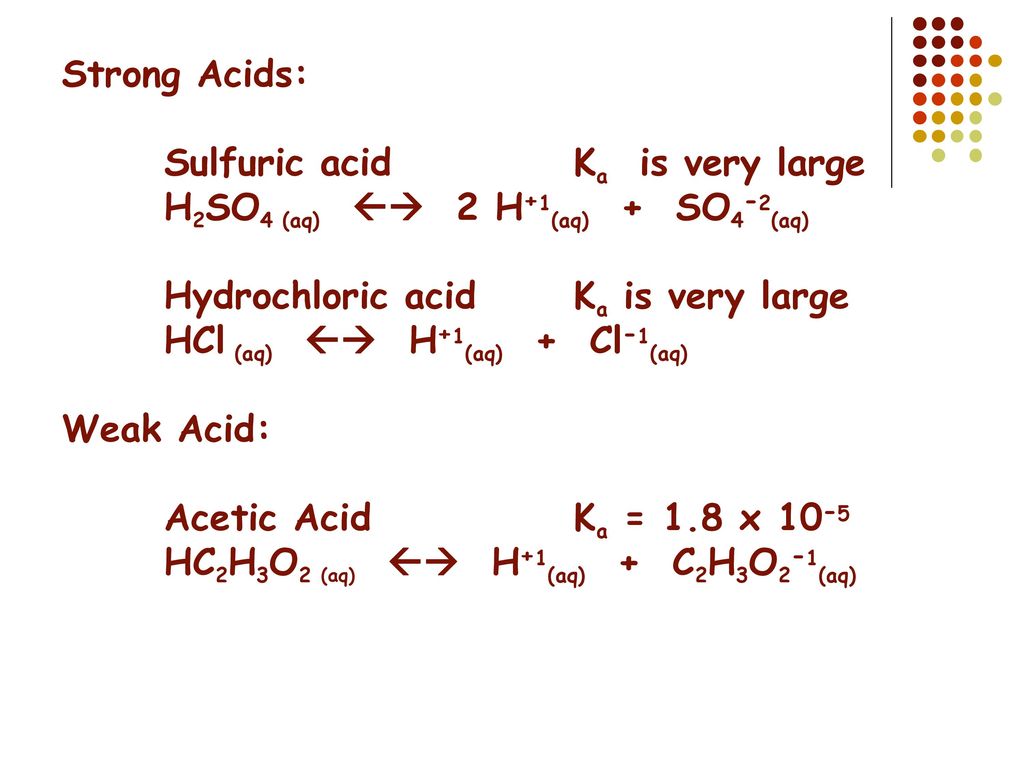
How much H2SO4 (in gram) must be added in 500ml of 0.1M CH3COOH (Ka=10 5) to change its degree of dissociation by 200 times
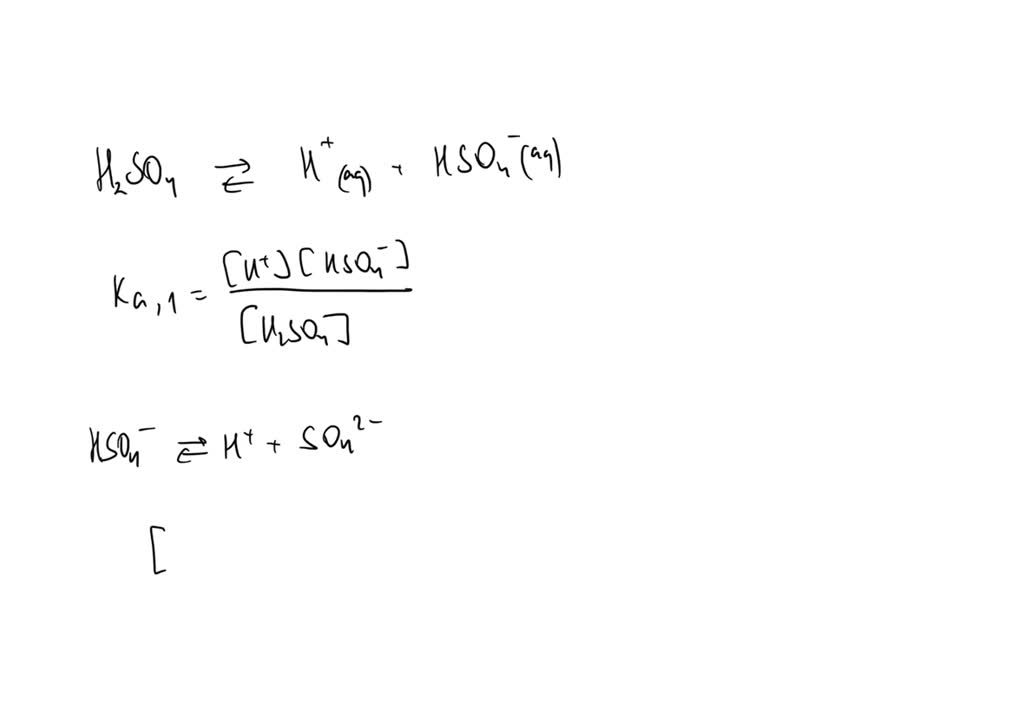
SOLVED: Write the acid dissociation equilibrium for H2SO4 in water and write the Ka expression. Is this a strong or weak acid? Are products, reactants, or both favored at equilibrium? Answer: This

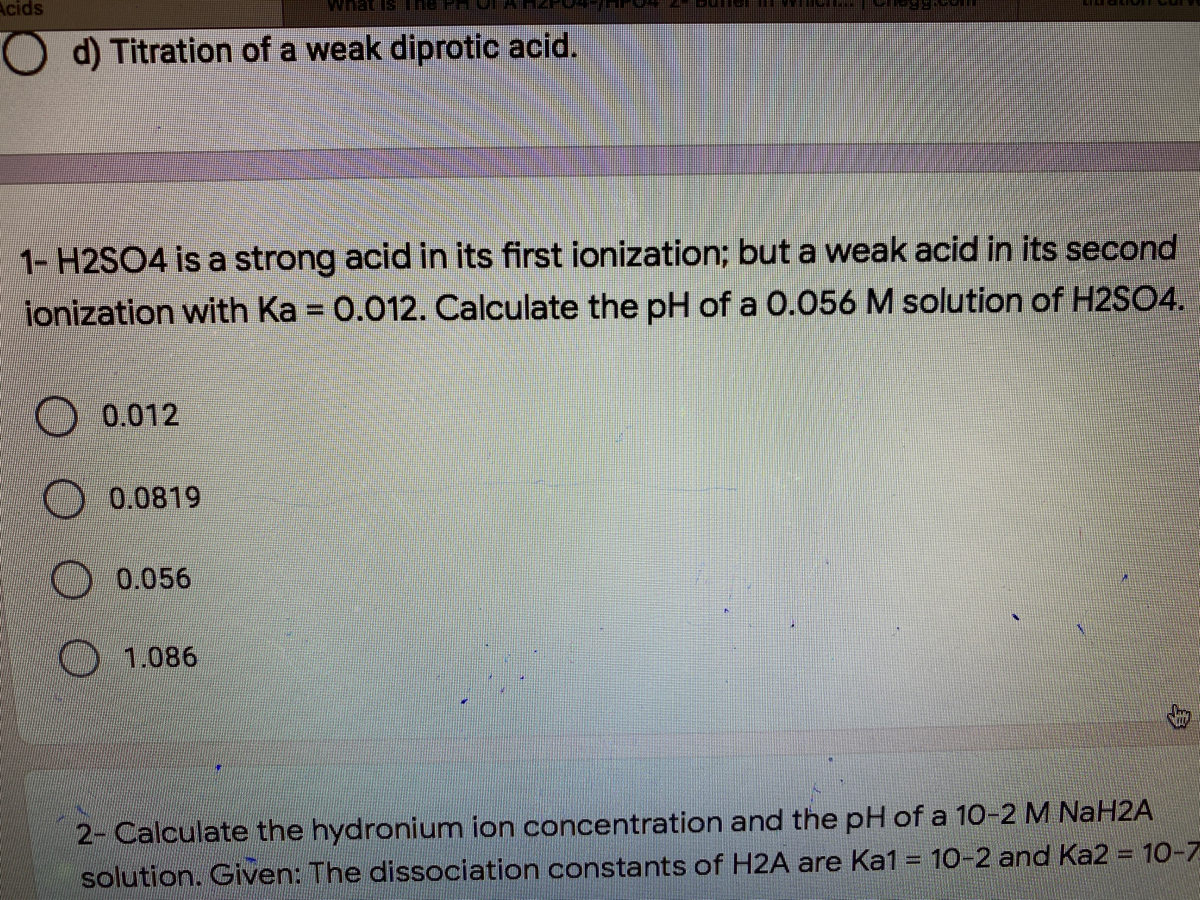
If Ka1 and Ka2 of sulphuric acid are 1 × 10^-2 and 1 × 10^-6 respectively, then concentration of sulphate ions in 0.01 MH2SO4 solution will be:

sir iss 3 question mei h2so4 ka Ph 0 7 kaise aagya kyuki h2so4 to diprotic hai isme two deprotanations - Chemistry - - 14691351 | Meritnation.com