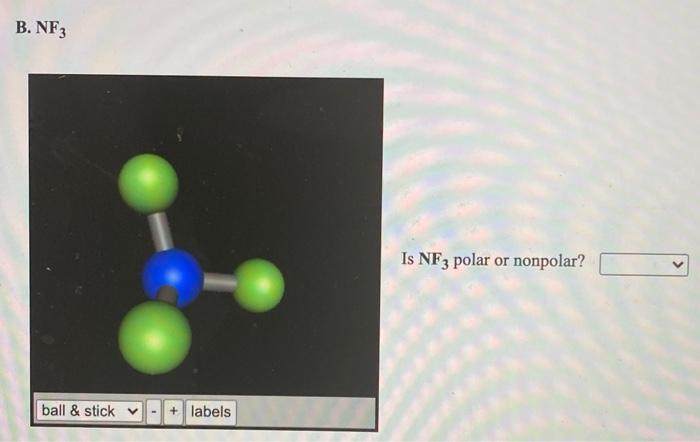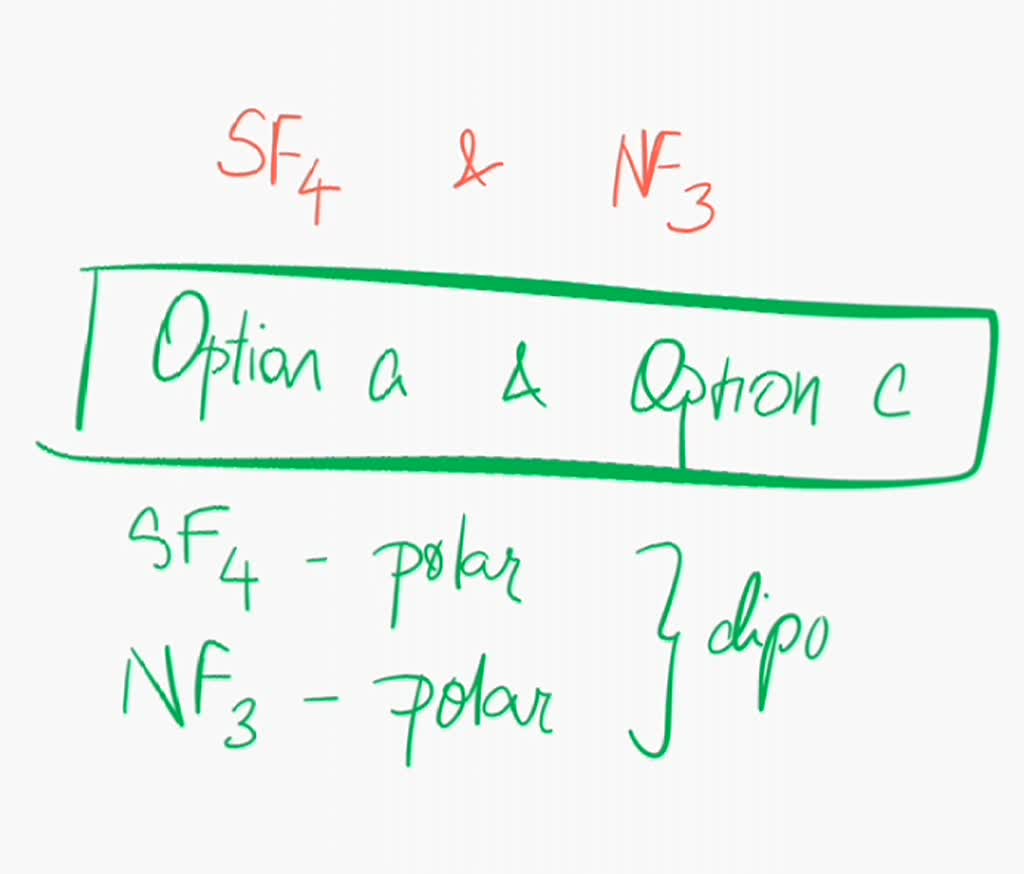
SOLVED: Are sulfur tetrafluoride (SF4) and nitrogen trifluoride (NF3) polar or nonpolar? (Select all that apply: ) SF4 is polar; SF4 is nonpolar: NF3 is polar; NF3 is nonpolar:

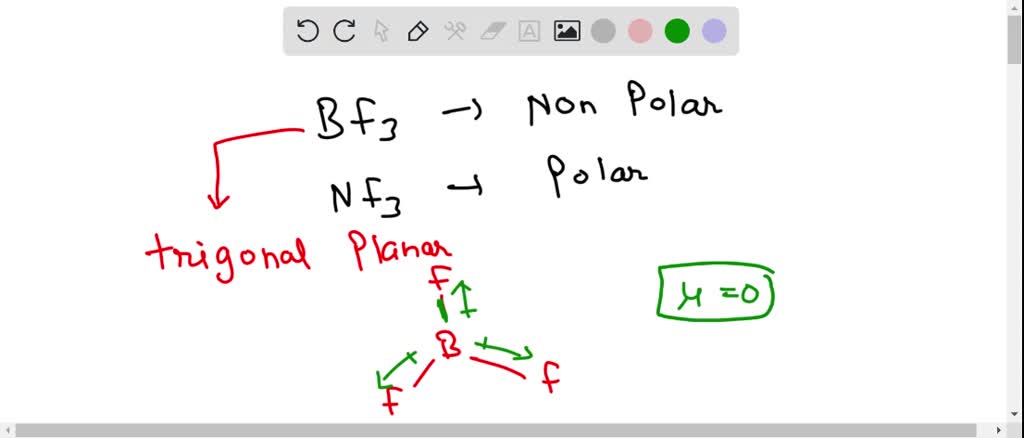
The molecule BF3 and NF3 , both are covalent compounds but BF3 is non - polar and NF3 is polar. The reason is that:

Which statement for NH3 and NF3 is false? Electro negativities N = 3.0, H = 2.1, F = 4.0 - Home Work Help - Learn CBSE Forum

Explain why nitrogen trifluoride has a small dipole moment even though it has polar bonds in a trigonal pyramidal arrangement. | Homework.Study.com

BF3 and NF3 both are covalent compounds but NF3 is polar whereas BF3 is non - polar. This is because :
Dipole moment of NF3 is less than that of NH3, even though N-F bond is more polar than N-H bond. Explain. - Sarthaks eConnect | Largest Online Education Community