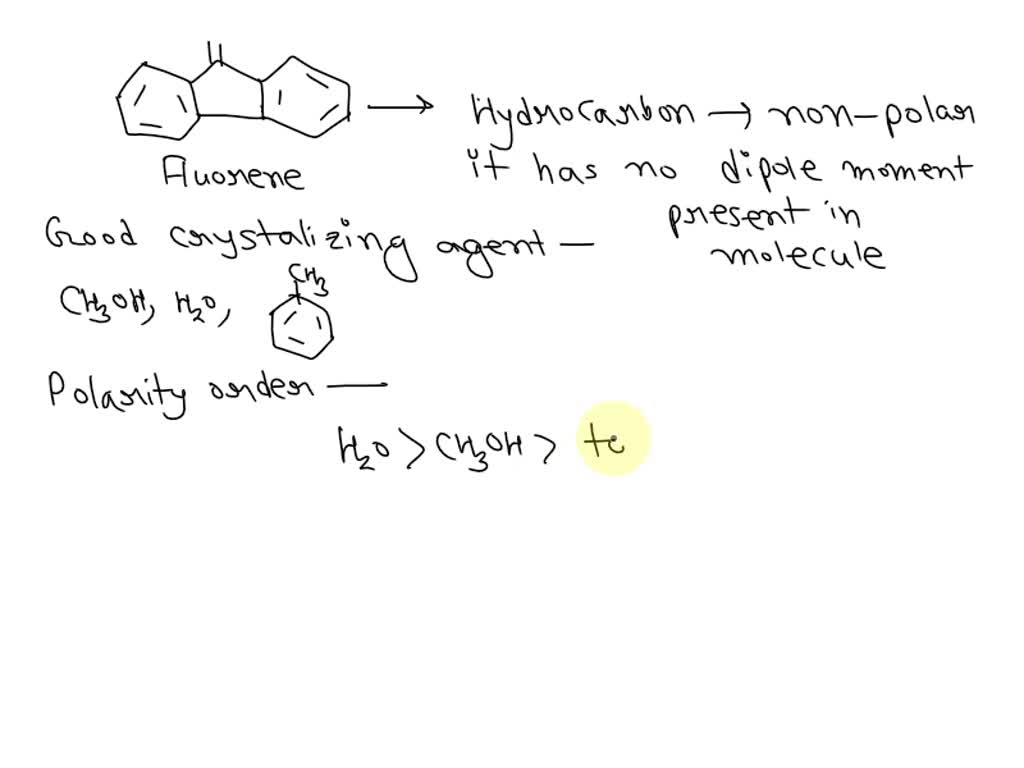
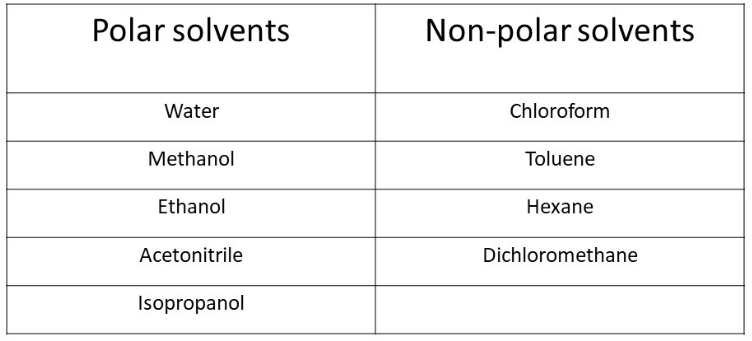
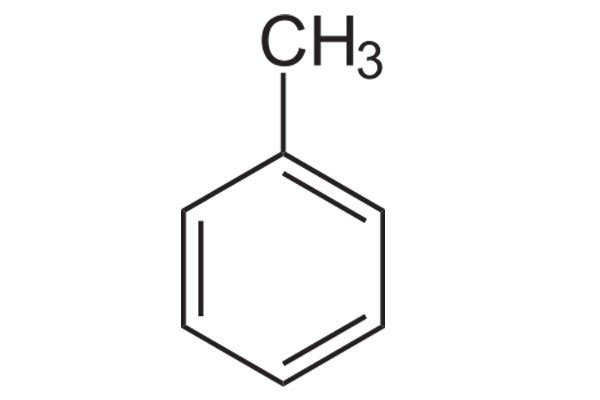
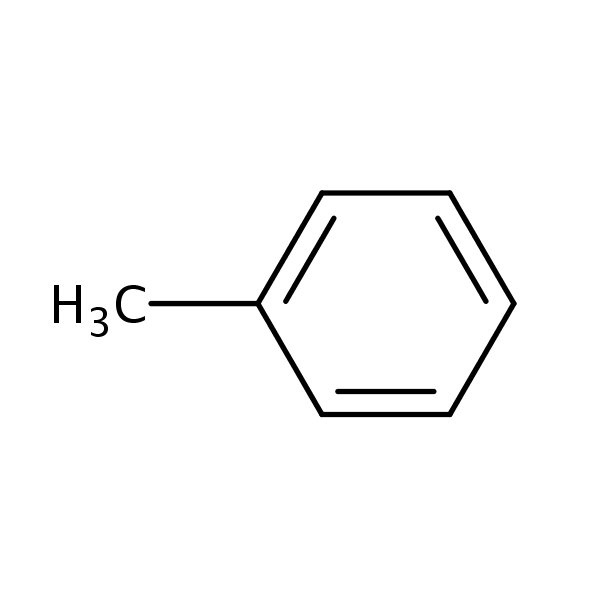
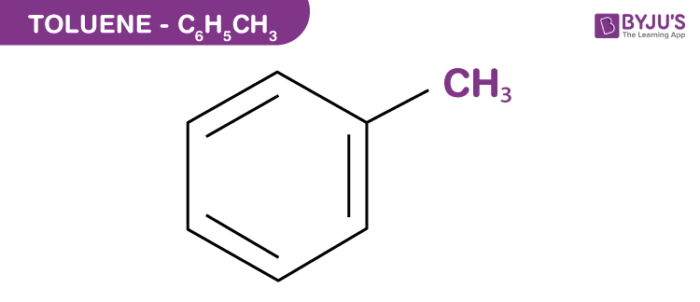
Arrange the following solvents in order of increasing polarity: a) ethanol b) ethyl acetate c) petroleum ether d) toluene e) acetone | Homework.Study.com
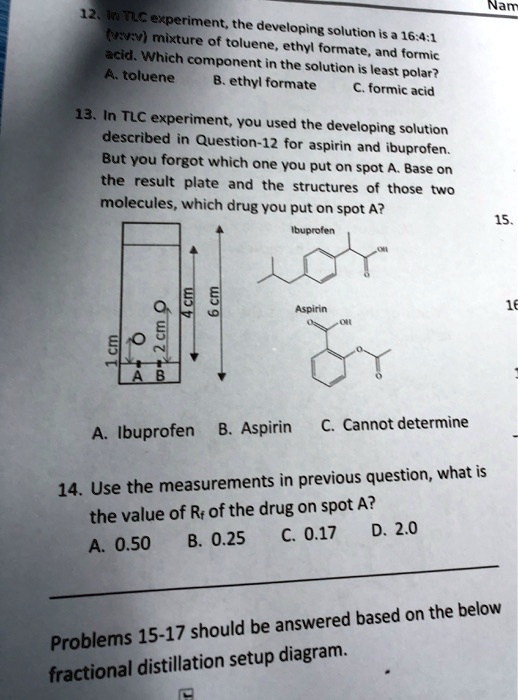
SOLVED: Veo Fs experiment; the developing solution (RRv) mixture of toluene 16.4.1 ethyl acetate, and formic. Which component in the solution is toluene least polar? ethyl acetate formic acid. In the TLC