NH3, PF3, and SF6 are all gases at room temperature. What intermolecular forces are present in liquid samples of each compound? | Homework.Study.com

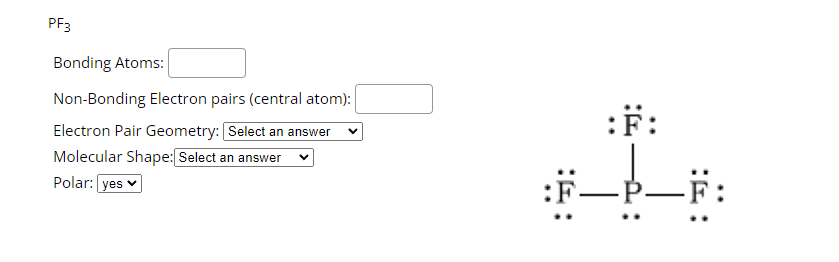
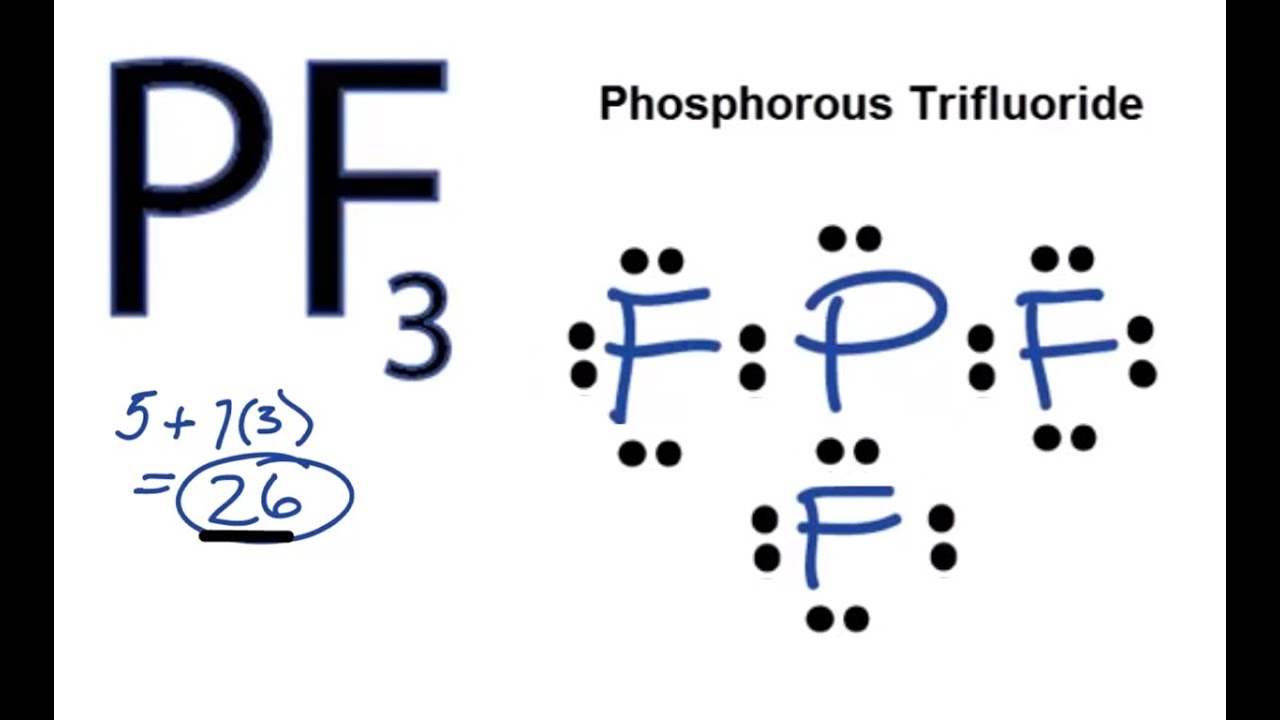
PF3 lewis structure, molecular geometry, electron geometry, polar or nonpolar, bond angle | Molecular geometry, Molecular shapes, Molecular