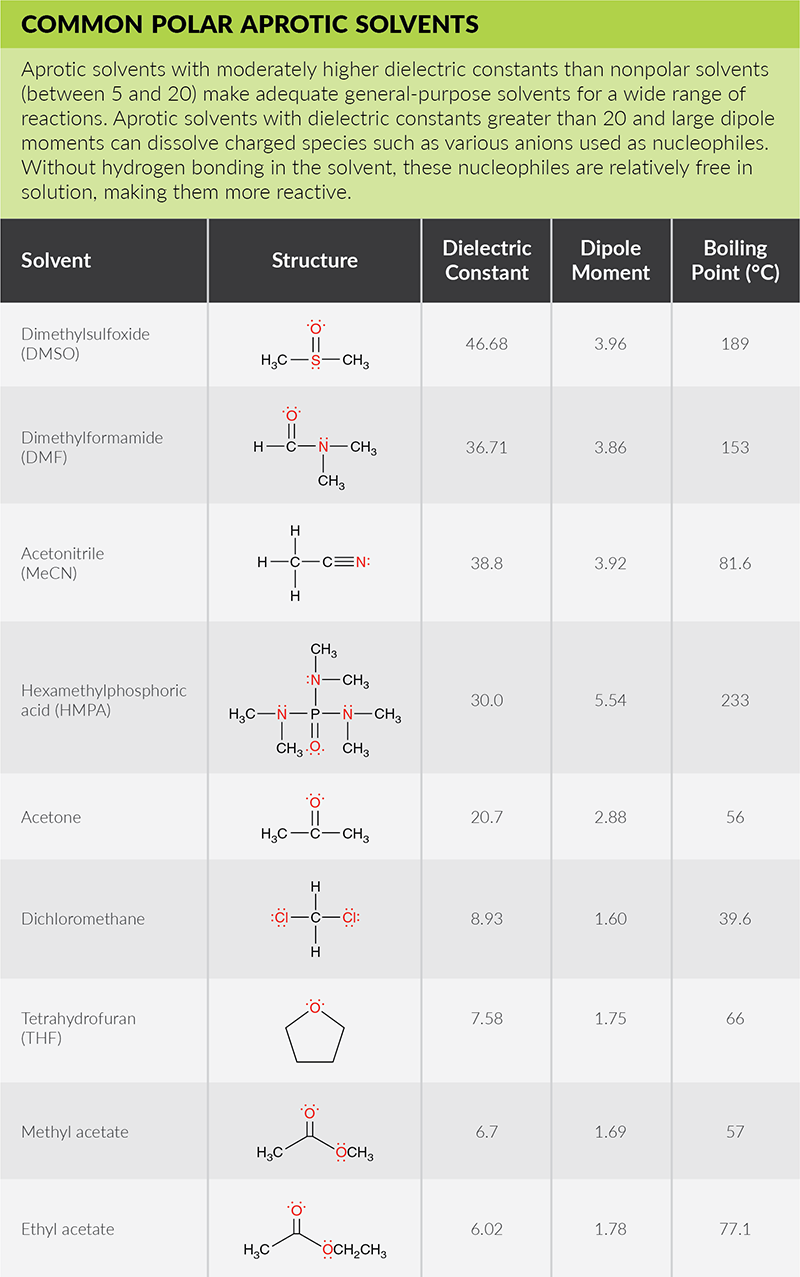
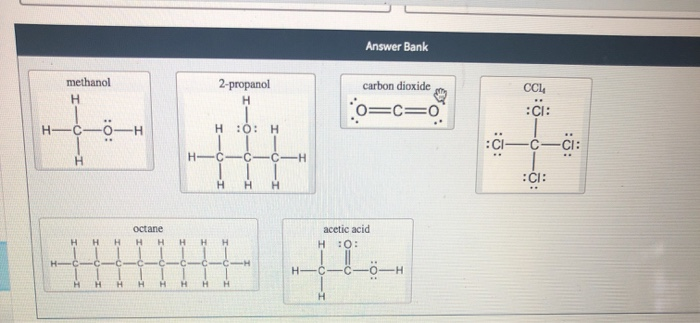
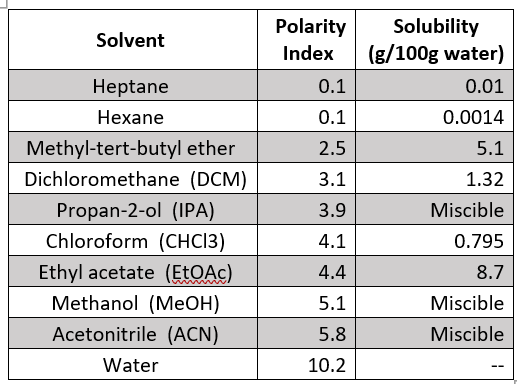
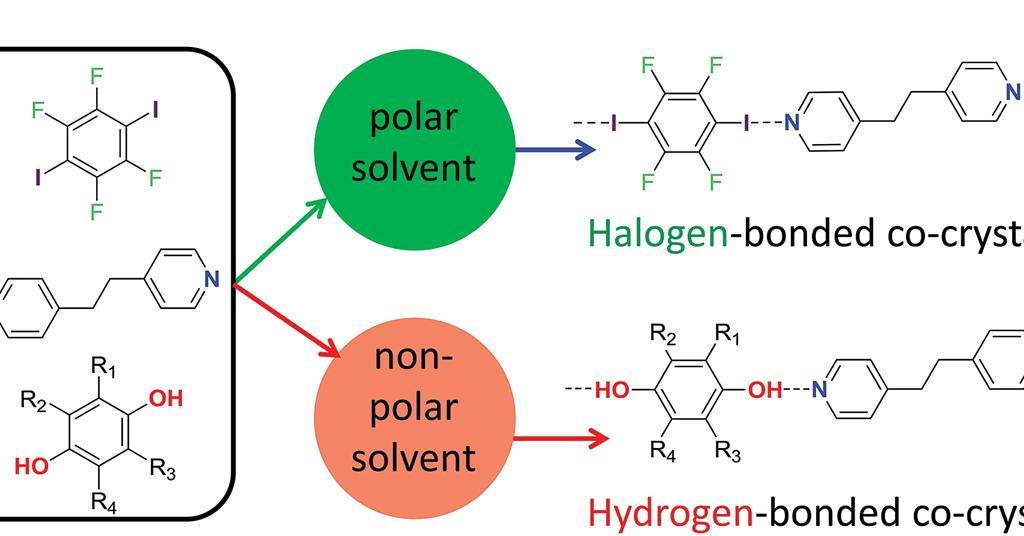
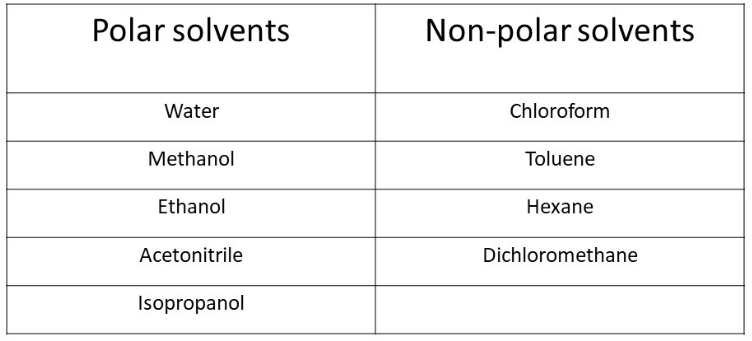
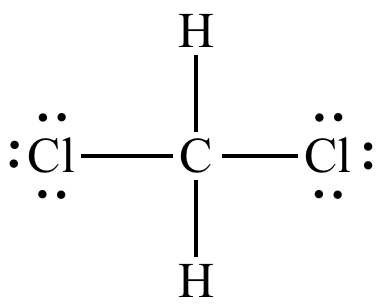
Polar solvent favours keto form while non polar solvents favours enol form. Which has the maximum enolic form in CCI4 (1) CH3COCH2COCH3 (2)CH3COCH2COH (3)CH3COCH2COOC2H5 (4)CH3COCH2CH3
Adding surfactants in non polar solvent, the micelles structure will look like - Sarthaks eConnect | Largest Online Education Community