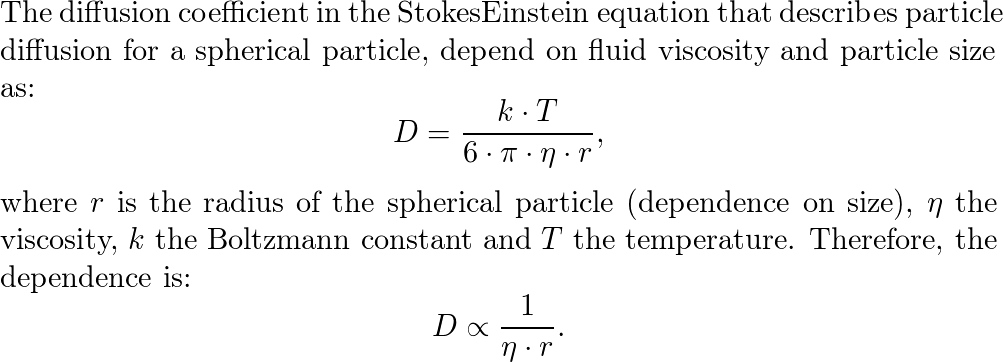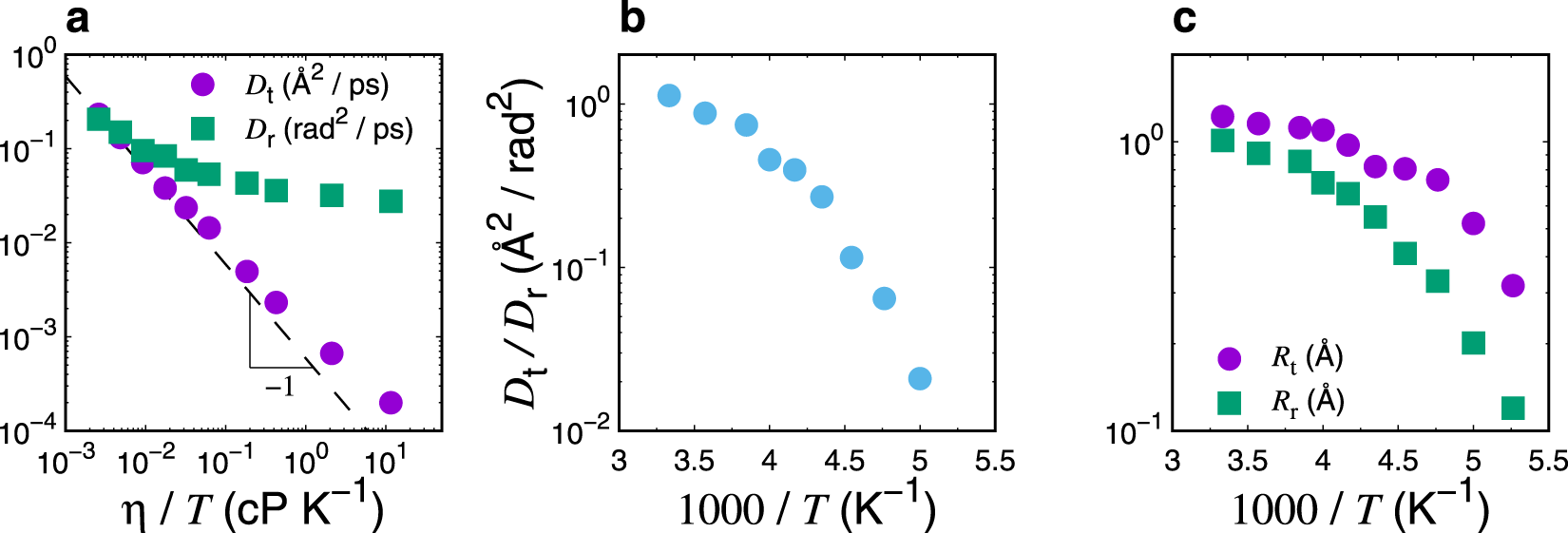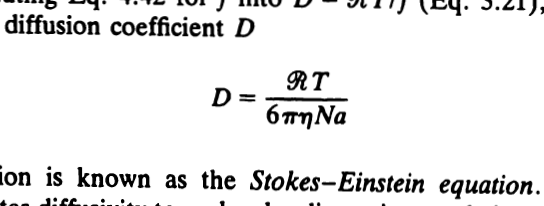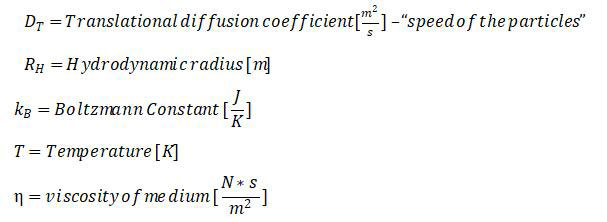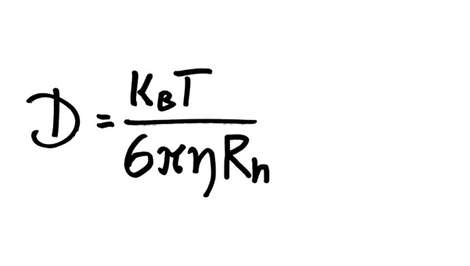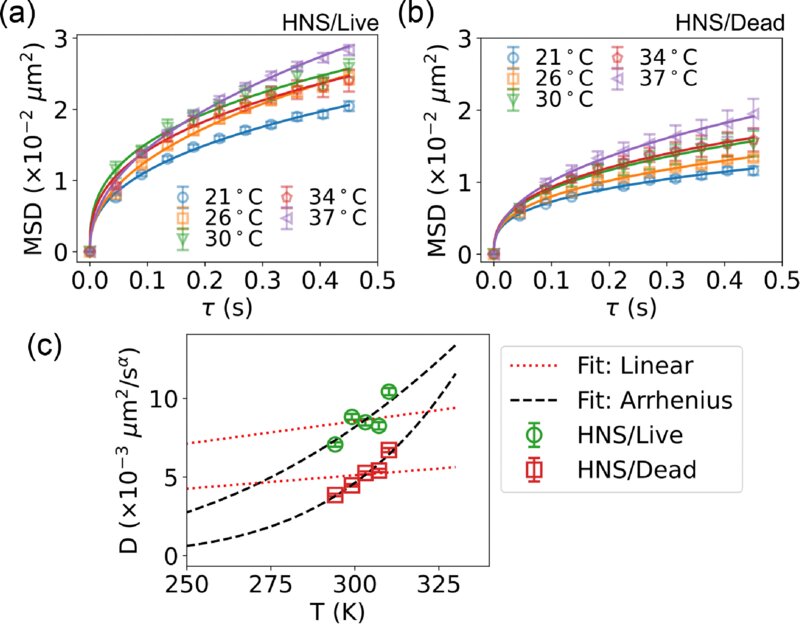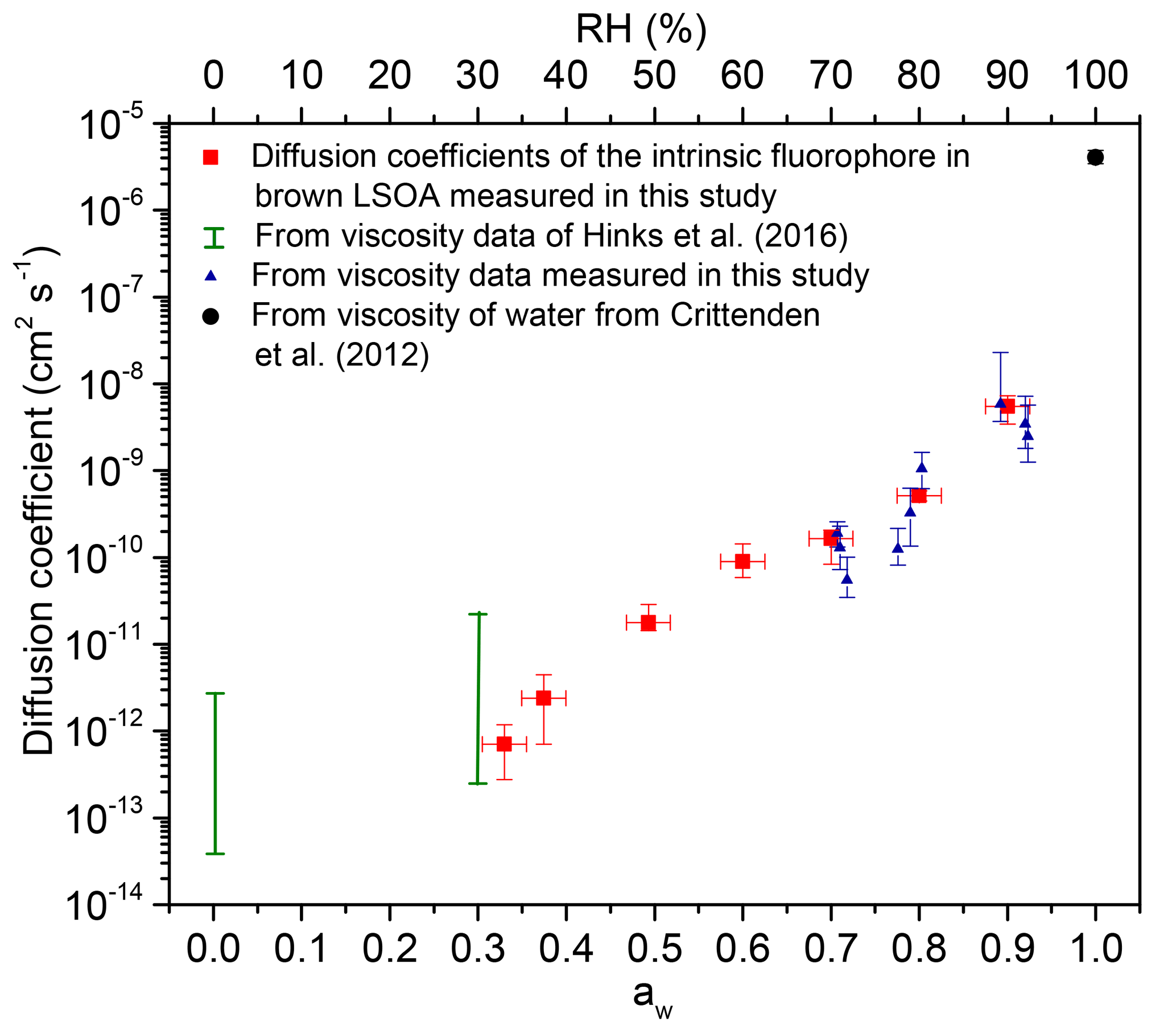
ACP - Viscosities, diffusion coefficients, and mixing times of intrinsic fluorescent organic molecules in brown limonene secondary organic aerosol and tests of the Stokes–Einstein equation
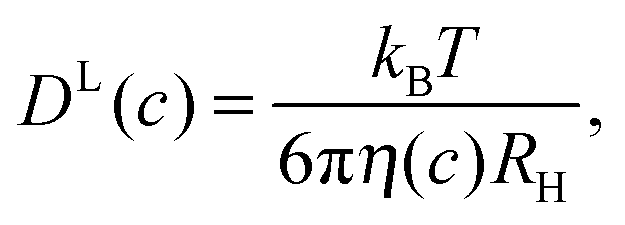
Transient binding accounts for apparent violation of the generalized Stokes– Einstein relation in crowded protein solutions - Physical Chemistry Chemical Physics (RSC Publishing) DOI:10.1039/C6CP01056C

PPT - Breakdown of Stokes-Einstein Relationship and pedal-like motion in s PowerPoint Presentation - ID:1442194

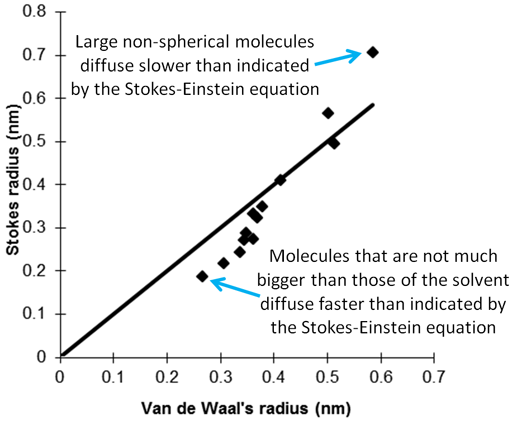
The Stokes-Einstein equation gives an estimate of the frictional coefficient, f, in terms of molecular

Fick's Law of Diffusion.pdf - Stokes-Einstein Equation D = Diffusion coefficient K = Boltzmanns constant T = absolute temperature K r = molecular | Course Hero
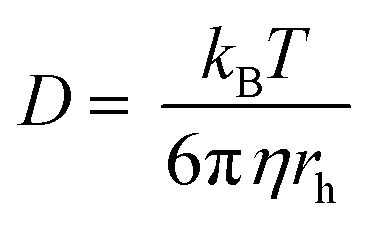
Estimating the shape and size of supramolecular assemblies by variable temperature diffusion ordered spectroscopy - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C4OB01373E
How would Einstein regard complex subjects like Fluid Mechanics and Continuum Mechanics? Why would he pursue abstract ideas and completely disregard unsolved problems of physics of daily life? - Quora

SOLVED: Using the Stokes-Einstein equation and the Wilkie-Chang correlation (eq: (6.6.25), see below), estimate the diffusion coefficient of oxygen in water at 298K. Take the molecular diameter to have a value of